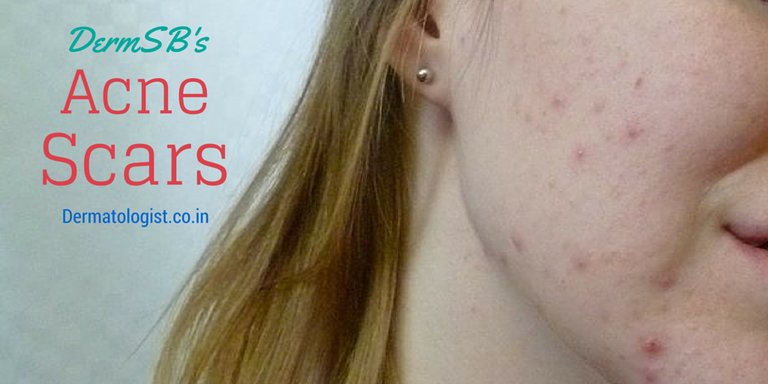

Though many cosmetic services claim that they can erase acne scars miraculously, acne scar is one of the most common and recalcitrant cosmetic dermatology problems. It affects 40-50 million people in United States alone. It is estimated that 95% of acne sufferers have some form of scarring.
The gold standard for scar treatment has been lasers. The effective ablative lasers are prone to side effects especially on pigmented Asian skin. The safe, non-ablative lasers promise collagen stimulation and like most others, the promise is hard to recreate on the patient’s face.
Fillers for acne scars is not a refreshingly new concept though FDA approval for the same has come only now. FDA recently approved Bellafill®, a filler developed by Suneva Medical Inc, for the treatment of moderate to severe acne scars on the cheeks of patients over the age of 21.
Bellafill® is composed of 80% denatured collagen and 20% PMMA with 0.3% lidocaine. PMMA and denatured collagen are hypo-immunogenic, and lidocaine will reduce the injection pain. The denatured collagen is processed without cross-linking or telopeptides to reduce immunogenicity.
The biggest drawback of a filler based acne scar correction is the need for repeated treatments. Bellafill® promises longer effect because of the PMMA beads stimulating endogenous collagen production. (a promise like most others!). However Bellafill® will definitely add a new dimension to acne scar treatment though Good Things Don’t Last Forever!!
- Machine learning-based BOTOX API - April 11, 2023
- Skinmesh: Machine learning for facial analysis - November 10, 2020
- Free Dermatology EMR for Machine Learning and Artificial Intelligence - January 2, 2020
this is the very nice and helpful information.
Thank You!
Many people face a lot of skin issues. In fact excessive hair on the face are the major problems faced by women and so Dermatologist in Indore helps in getting rid of such problems.